|
Directions: Now that you have learned more about what happens in a chemical change, you are going to create a new model of what happened in the instant cold pack we saw at the beginning of this unit. Make sure you include the following:
Reactants
Products
0 Comments
Directions:
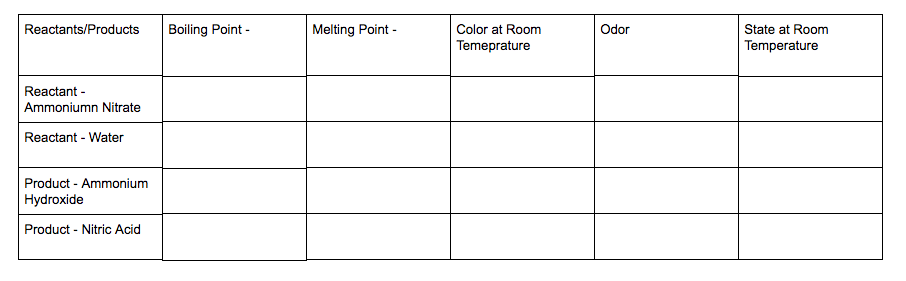
1. Copy the table below into your journal. Title the page "Properties of Reactants and Products." 2. Conduct research in this table to learn about the properties of our reactants and the products they make in the instant cold pack. 3. When you are finished, answer the following questions in your lab notebook underneath your table. Please restate the question as part of your answer. a. What do you observe about the properties of our reactants and products? b. The reactants and products have very different properties. Why do you think that is? Think about what you know about the atoms. c. Come up with another rule for chemical change regarding the properties of reactants and products. 4. When you are done and checked in, click "Read More." Add any new information to your explanations. Directions:
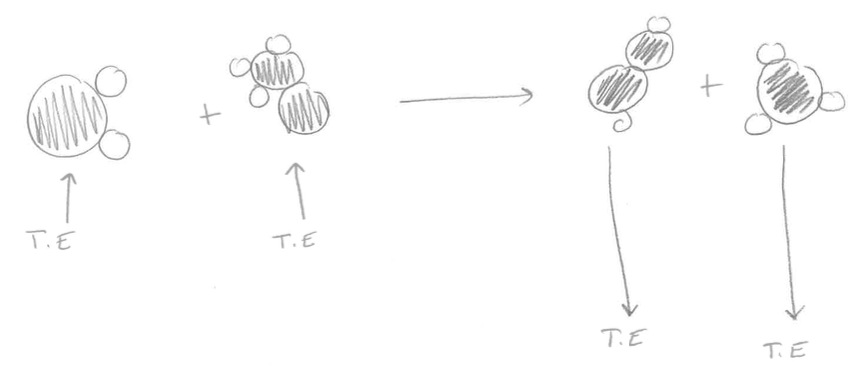
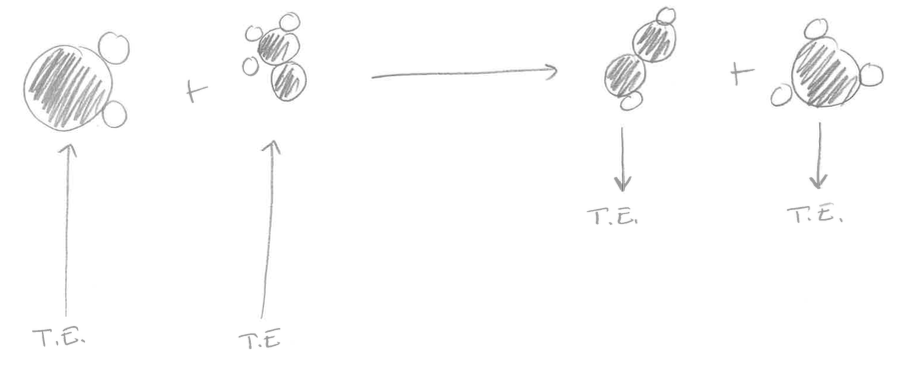
You are going to create a model of the endothermic and exothermic reactions we saw in the lab (baking soda + vinegar; elephant toothpaste). You need to show what is happening to the reactant molecules and how energy is released and absorbed to create the products. Here are the equations for each of those reactions. Baking Soda and Vinegar C2H4O2 + NaHCO3 ----> NaC2H3O2 + H2O + CO2 Elephant Toothpaste 2 H2O2 ----> 2H2O + O2 Remember to show what is happening with the molecules and energy associated with breaking and reforming bonds. Here is the link to the animations we watched to help you! Directions: 1. Divide your page in half down the middle. Label the left side "My first thoughts" and the right side "My new thoughts. Title the page "Endothermic vs. Exothermic." 2. Look at the drawings of chemical reactions below. Explain which one is an endothermic reaction and which one is an exothermic reaction and why. Use your understanding of reactants, products, chemical bonds, thermal energy, and atoms in your answer. Use you notes in your journal to help you. 3. When you have checked in with a teacher, click "Read More" and write your new thoughts in the right hand column. Picture #1 Picture #2
Directions:
I have included the link to the animation of molecules in a reaction that you saw in our discussion to help you out! CLICK HERE for the link! Huzzah!!! Directions for when you are finished all three levels!
Once you have finished balancing all of the equations, you need to do the following.
Directions:
1. Create a double entry. Split your page down the middle. Label the left side "My First Thoughts" and the right side "My New Thoughts." Title the page "Counting Atoms in Reactants and Products." 2. Look at the table below. Explain what is wrong with it AND WHY. Also explain what you would have to do to make it more accurate. Include your understanding about what happens to molecules in chemical reactions to support your answer. 3. Click "Read More." to see what is more accurate and why Using your new knowledge about chemical change and reactants and products, create a model of what happened when we activated the instant cold pack. The information you need on the reactants and products is below. Think about what happened in the Alka-Seltzer and Water reaction as well as the methane and oxygen reaction to help you with your model (I have provided a link here to help you if you need it)
Instant Cold Pack Reactants
Products
When you are finished with your model and have checked in with a teacher, please post it to Seesaw. Then, reflect in your Running Record on the "M&M Chemical Reaction" activity. Directions:
1. Create a double entry on your next blank page. Split your page down the middle. Label the left side, "My first thoughts" and the right side "My new thoughts." Title the page "Atoms and Molecules" 2. On the left side of your entry, draw each molecule below. This should be a review, but you can use the notes you have in your journal to help you. Explain each of your molecules to the best of your ability. 3. After you have checked in with Ms. Saikin or Miss Stahl, you may click, "Read More." |
NGSS Performance ExpectationsMS-PS1-2.Analyze and interpret data on the properties of substances before and after the substances interact to determine if a chemical reaction has occurred. |
Ms. Saikin's 6th Grade Science